For a long time, a lot of classroom explanations of magnetism mostly stayed with two categories: ferromagnets and antiferromagnets. Ferromagnets are the kind most people recognize right away. In those materials, many atomic magnetic moments line up in the same direction, which creates an overall magnetic field. Iron is the usual example. Refrigerator magnets and many common permanent magnets depend on that type of behavior.
Antiferromagnets work another way. Their magnetic moments point in opposite directions in an alternating pattern, so they largely cancel out. Because of that, they usually do not show the obvious outside pull people expect from a magnet. If someone handled one, it would probably not seem very magnetic at first.
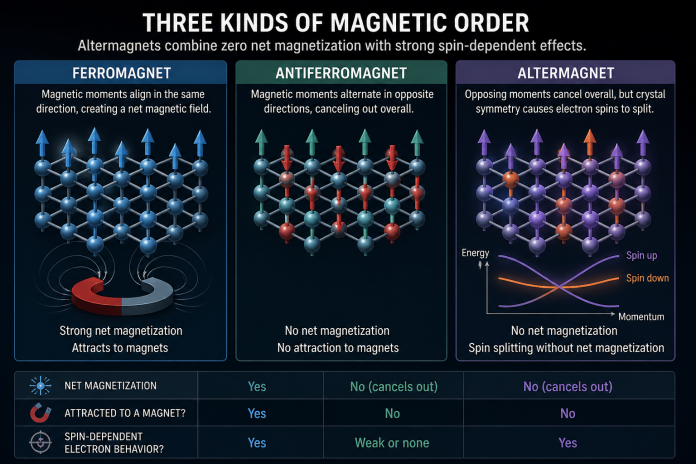
Researchers have been giving more attention to a third class called altermagnets.
They do not fit cleanly into the older two-part picture. Like antiferromagnets, they can have opposing magnetic moments that leave little or no net magnetization. From the outside, they may look fairly unremarkable. They are not the kind of materials that snap onto a filing cabinet or cling to a refrigerator door.
The interest comes from what happens inside the crystal. Electrons in altermagnets can behave in ways that resemble ferromagnets, especially when spin affects their energy states. That mix of traits was part of what drew researchers in. It did not match the simpler categories very well.
Crystal structure matters here. In solids, atoms sit in repeating patterns, and those patterns influence how electrons move through a material. They also affect how electron spins respond. Spin is a quantum property connected to magnetism, though the name can make it sound more mechanical than it really is.
In many standard antiferromagnets, opposite magnetic states remain electronically equivalent. In altermagnets, certain crystal symmetries can break that equivalence. Electrons with opposite spins may occupy different energy states even while the total magnetization stays close to zero. That is one of the main things scientists look for when identifying the class.
Researchers often describe this as spin splitting without net magnetization. It sounds technical because it is technical. The shorter version is that the material may not create a strong external magnetic field, but spin still changes how electrons behave in measurable ways.
That has drawn interest from the field of spintronics, which studies how electron spin could be used in memory devices, sensors, and computing systems. Ferromagnets already play a role in hard drives and some magnetic memory technologies. They also create stray magnetic fields, which can interfere with nearby parts when devices become smaller and more crowded.
Antiferromagnets avoid much of that stray-field issue, but they can be harder to detect or control. That has made them appealing in some designs and frustrating in others.
Altermagnets may offer a middle option. They can combine low net magnetization with useful spin-related effects. Researchers are studying whether that could help with faster memory, lower-power components, or improved sensors. It is still early, and there is no guarantee every proposed use will work out.
Many altermagnetic materials were already known before the label became common. Some compounds had been studied for years under other classifications. Later work suggested they belonged in a separate group instead. Science does this sometimes. Better tools or better theory can change how an older material is understood.
One of the best-known examples is manganese telluride, or MnTe. In 2024, researchers reported evidence that MnTe showed the kind of spin-split electronic structure predicted for altermagnets while still having almost zero net magnetization. Later studies examined ways to image and control altermagnetic order in thin films.
Magnetism ends up being less tidy than the first version many students learn. It is not always everything lined up in one direction or everything canceled neatly in pairs. Crystal geometry can matter a lot. So can electron interactions. Even a small shift in atomic arrangement may change how a material behaves.
This image is the property of The New Dispatch LLC and is not licenseable for external use without explicit written permission.





